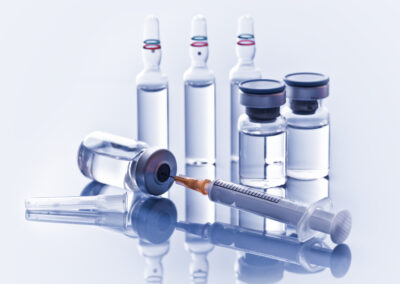
Visual inspections ensure that visible particulates are not present in products manufactured by an organization. ...
In pharmaceutical manufacturing, there is the need to use sterile water in production and cleaning activities. ...
Non-prescription drug products are classified as over-the-counter (OTC) drug products. OCT products are drugs that ...
A combination product is a product composed of two or more regulated components. Examples of combination products ...
Unapproved prescription drugs propose a significant risk for patients since they haven’t been reviewed for ...
Laboratories, whether clinical or non-clinical, have to follow certain Good Laboratory Practices (GLP). ...
When an event occurs related to a medical device there is the need to determine if it qualifies as an adverse ...
In 1976, the Medical Device Amendments of the Federal Food, Drug, and Cosmetic Acts established three regulatory ...
There are many different types of Regulatory audits that can be conducted. These are audits that upon successful ...
A certificate of analysis (CoA) is a document that manufacturers create that verifies the products they ...
Quality is something that should be integrated into an organization. To pursue quality means to drive forward ...
Accelerated stability testing increases the rate of chemical degradation and physical change of a drug by using ...